“Together, we Trial wisely.”
Characterised by high pressure and even higher stakes, clinical trials are environments in which business success is overshadowed only by the prospect of advancing the state of human health. These lifechanging studies must be kept moving forward, delivered through systems designed to drive confident decisions and calm action. Global technology firm Suvoda has stepped up to the plate in an increasingly complex industry, delivering a market-leading software platform equipped to support the most critical moments in a trial. We heard more from Robin Abadía below, Director of External Communications at Suvoda, as the company is named in the Global Excellence Awards 2026.
For over a decade, Suvoda has been on a mission to advance human health by streamlining the clinical trial experience for patients, sites, sponsors, and CROs alike. To do so, it has developed a patented, real-time software platform is purpose-built for the most complex, mission-critical, and time-sensitive moments in studies. This platform empowers people to make confident decisions and take calm action whilst operating within high-stakes studies.
Suvoda was founded in 2013 by CEO Jagath Wanninayake and COO Robert Hummel, two experts in eClinical technologies who were driven by a desire to ease the complexities faced by clinical trial participants and professionals. Jagath’s background in clinical trial technology, which also includes previous experience as the Co-Founder of Clarix LLC, has laid the foundation for Suvoda’s innovative approach to clinical trials and has since propelled the team to significant success.
The pioneering Suvoda Platform provides a suite of eight products on a single, patented ecosystem: IRT, eCOA, eConsent, Greephire Patient Payments, Greenphire Travel, Scheduling, Site Payments, and Budgeting and Benchmarking. This fully integrated model minimises friction and maximises engagement for sponsors, CROs, sites, and patients, not only simplifying but enhancing the experience and contributions of all users across the clinical journey.
“We can support even the most time-sensitive, mission-critical points of a trial, so that life-changing studies keep moving forward and everyone involved can ‘trial wisely’.”
“Some other clinical trail technology companies will also use the term ‘platform’,” said Robin, “but what makes ours unique is that the Suvoda Platform isn’t a variety of separate systems simply ‘integrated’ at the API level; ours is unified as a cohesive system. Because we are a disrupter in the industry, our patented, modern back-end architecture allows the front-end user’s experience to feel significantly improved.”
Using the Suvoda Platform, study teams benefit from a single log-in, a unified data layer, and an intuitive interface that was created with user-centricity in mind. Beyond the technology itself, the company’s exceptionally prepared client services teams support sponsors and CROs for the full lifespan of each trial, providing protocol-level expertise, calm guidance, and global support.
Since its launch, Suvoda has been chosen to support sponsors and CROs in more than 2,000 trials across over 95 countries, including many of the most complex and high-stakes studies in oncology and rare disease. Meticulously monitoring customer satisfaction and feedback, the company consistently achieves scores that exceed typical technology industry benchmarks, reflecting the trust that clients have placed in its platform and services. External validation, from industry awards and analyst reports to strategic partnerships and growing adoption, have further evidenced that Suvoda is meaningfully enhancing the clinical trial journey.
“We pride ourselves on being deeply patient- and site-centric, designing digital experiences that not only streamline trial operations but also reduce friction for the people actually moving through trials,” Robin told us. “People now expect, and deserve, to have technology that just works. That’s especially needed for the high-stakes world of clinical trials. We’re proud to be seen as an innovator and leader in our space, with five U.S. patents for our platform architecture and independent recognition from analysts for our impact on clinical trial technology.”
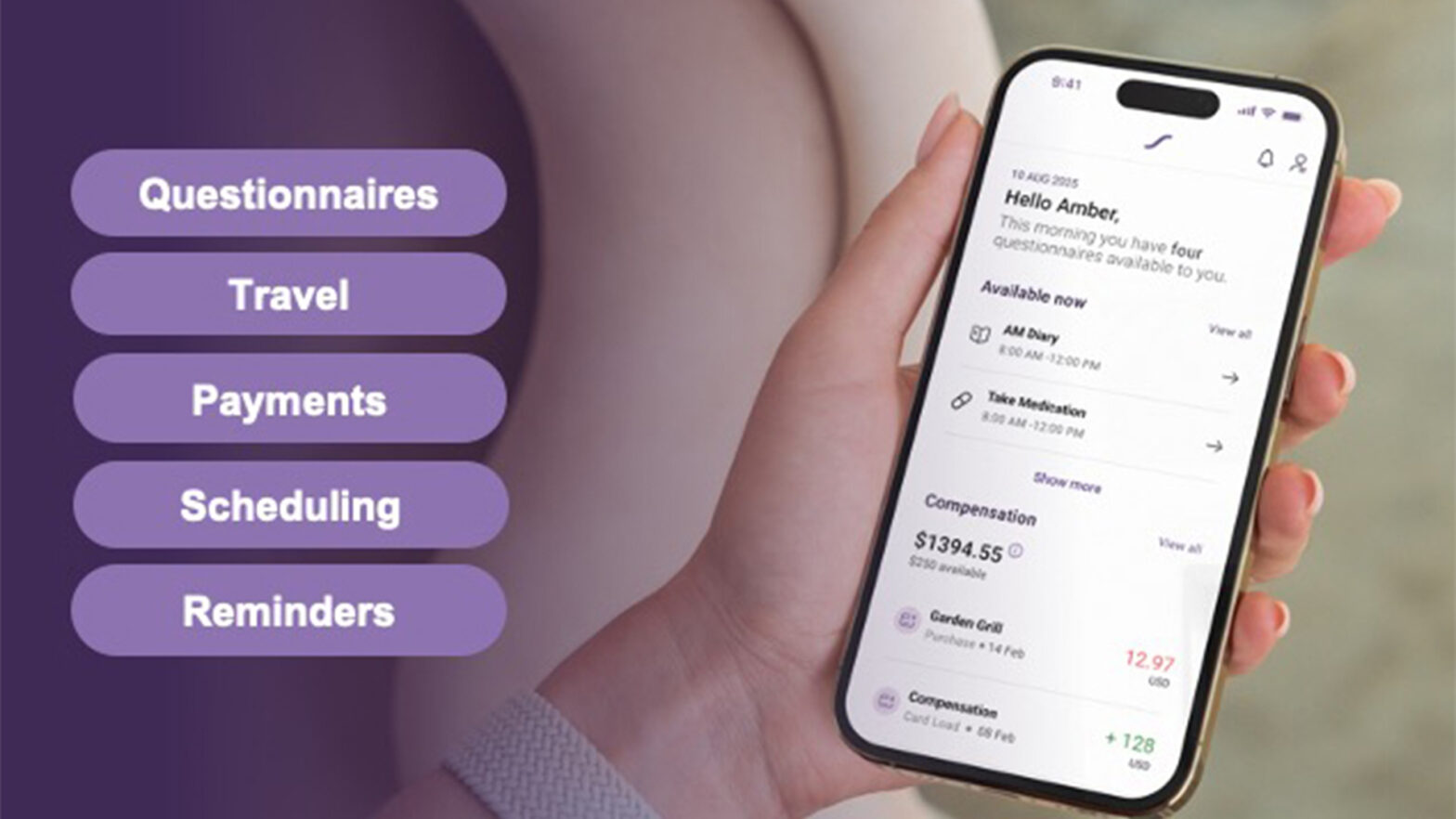
Building on its exceptional success, Suvoda has developed a groundbreaking patient app. The Suvoda patient app provides trial participants with a single, intuitive mobile experience that allows them to manage the essentials of their trial needs in one unified place. On their own device, users can access visit reminders and see their scheduling, complete eCOA questionnaires and fill out daily diaries, arrange travel details, and track patient payments and reimbursements, all without the unnecessary burden of juggling emails, calls, and papers.
The bright minds at Suvoda have also created Sofia, an AI assistant designed for Suvoda IRT, the company’s software for patient randomisation and trial drug supply management. Sofia transforms what used to be multi-screen, multi-click workflows into simple, auditable conversations. With Sofia, Suvoda aims to help study teams quickly access trial data, reports, and documentation through a secure, streamlined chat interface.
Together, these innovations enable Suvoda to further unify the digital experience for patients while simplifying day-to-day work for sites and sponsors. With all features comprised in a sole, user-friendly interface, the patient app promises higher patient engagement, fewer missed visits, and better participant retention. With many administrative tasks now streamlined into a guided, self-service experience for patients, site burden is reduced as staff are free to shift their focus from coordination to clinical care.
Furthermore, study teams can make faster and more confident decisions with support from Sofia. Without compromising role-based access or blinding, the AI technology can surface information such as inventory levels, shipment, or visit data; create tables and charts; and link directly to relevant sections of user manuals. Powered by the same unified Suvoda Platform, these innovations make for a more connected ecosystem overall, allowing data and workflows to move smoothly between patient, site, and sponsor tools.
Of course, as with many industries, one of the biggest developments on everyone’s mind is the use of AI. The technology has significant potential, but there is absolutely no room for error in the field of clinical trials, when dealing with patient’s health or the carefully designed protocols of a clinical trial study. With this understanding, Suvoda’s approach to AI is innovation with guardrails: every AI use case must be reliable, secure, and fully auditable.
This can bee seen most clearly in Sofia. The AI agent is designed to only answer questions it has been taught to answer using expert-authored instructions, has been validated by humans, and operates within strict role-based access that maintains study blind. What’s more, with “zero-day” data retention, Sofia never exposes data beyond a user’s permissions and never uses customer data to train public models.
“AI brings both opportunities and challenges to our industry,” Robin stated. “Sponsors and sites want faster, simpler ways to work, but they’re rightly cautious about AI risk. We’ve turned that tension into an advantage by harnessing AI in ways that support our users while also preserving the precision that our industry demands.”
In the coming years, Suvoda will continue to combine innovation with expertise so that it can support end users whilst responding directly to the needs of the industry. In alignment with this mission, the company is excited to deliver on its promise of the Suvoda-Greenphire merger announced last year. By uniting its central nervous system for trials – comprising randomisation, drug supply, consent, outcome data, and eCOA – with Greenphire’s strength in patient payments, patient travel, budgeting, and site payments, Suvoda is making significant strides in building a truly end-to-end platform that serves the needs of clinical trial sponsors.
Through 2026 and beyond, Suvoda seeks to help sponsors move away from fragmented point solutions and transition to its unified suite of solutions. The benefits are myriad: patients and sites can experience a single, intuitive platform and Suvoda app that handles their needs; sites deal with fewer systems and less manual reconciliation so they can focus on care; while sponsors and CROs gain a trusted partner providing connected clinical and financial data, allowing them to see what is happening across enrolment, logistics, and budgets, and act faster.
Ultimately, Suvoda’s unified tech suite opens up workflow improvements that the healthcare sector has been long overdue. Through its pioneering platform, patient-preferred app, and complementing AI assistant, the company has successfully developed a comprehensive ecosystem that streamlines the clinical trial process for all involved. With plans for further innovation already underway, Suvoda is gearing up for another year of making meaningful impact on the state of human healthcare, a mission that has earned it recognition as the Most Innovative Clinical Trial Technology Firm 2026.
Contact Details
Contact: Robin Abadia
Company: Suvoda
Web Address: www.suvoda.com